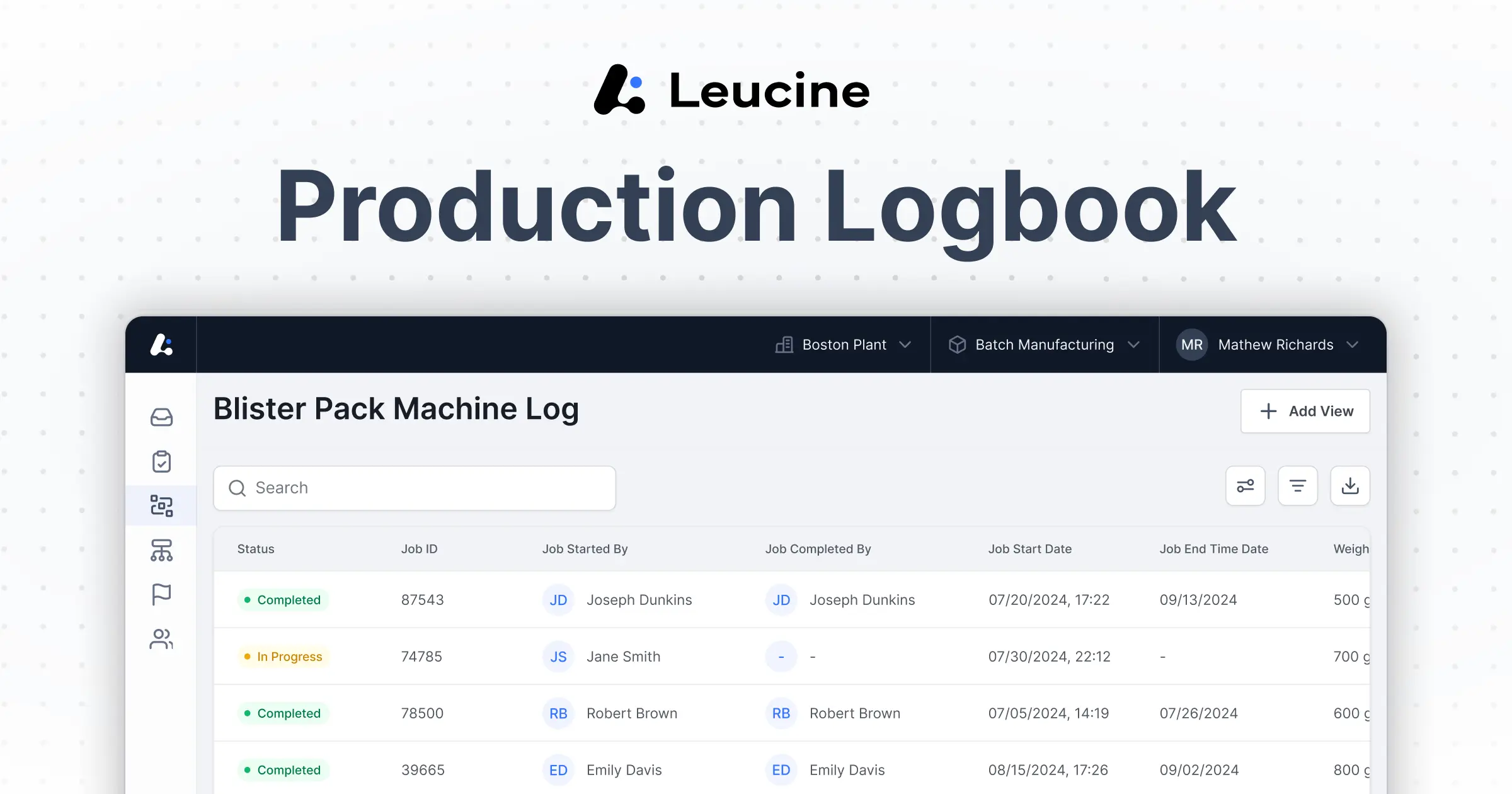
Standardize Workflows, Ensure Data Integrity With AI-Powered Electronic Logbook Software.
Ensure every batch is delivered On Time, In Full (OTIF) with AI-powered elogbook software—achieve flawless execution and real-time visibility.





















MES-Integrated Electronic Logbook
Software for a Single Source of Truth
Capture data in real time, ensure compliance, and streamline shop floor workflows
with MES-connected logbook software.
Software for a Single Source of Truth
with MES-connected logbook software.
Reduce Documentation Errors
Eliminate manual entries with automated records. Ensure accuracy, minimize data entry mistakes, and lower compliance risks.
Prevent Production Bottlenecks
Get real-time visibility into shop floor operations. Detect risks early and resolve issues to prevent recurring failures.
Reduce Operator Effort
Auto-capture timestamps and integrate with systems like SAP. Cut down manual entry and simplify operator tasks.

Connect logbooks with batch execution, quality, and maintenance systems.
Digitize Logs using AI.


Standardize shop floor workflows.
Prevent missing steps. Enforce SOP compliance with Process Interlocks.


Capture Data Efficiently
Always audit-ready, Reduce inspection risks.

Frequently Asked Questions
1. What is Leucine Production Logbook, and how is it different from other elogbook software?
Leucine Production Logbook is an AI-powered digital logbook software for pharma manufacturing. Unlike basic elogbook software, it integrates with MES, ERP, and QMS to automate data capture, reduce errors, and simplify compliance.
2. How does Leucine Production Logbook ensure data security and integrity?
Every entry is protected with electronic signatures, timestamps, and secure authentication. Leucine enforces ALCOA principles and complies with 21 CFR Part 11, GMP, and global regulatory standards.
3. Can Leucine Production Logbook integrate with existing MES, ERP, or QMS systems?
Yes. The platform provides open APIs and configurable connectors, enabling seamless integration with MES, QMS, and ERP—ensuring a single source of truth across all shop floor workflows.
4. How does real-time visibility in the logbook software improve manufacturing performance?
By monitoring workflows and capturing data in real time, Leucine identifies bottlenecks early, recommends corrective actions, and helps teams consistently achieve On Time, In Full (OTIF) delivery.
5. How does the digital logbook software support audits and compliance?
All log entries, edits, and approvals are automatically time-stamped, creating a secure, tamper-proof audit trail that simplifies inspections and ensures audit readiness for FDA, GMP, and global standards.
View and learn more about MES with our comprehensive list of resources













.png)
.png)
.png)


.png)
.png)
.png)